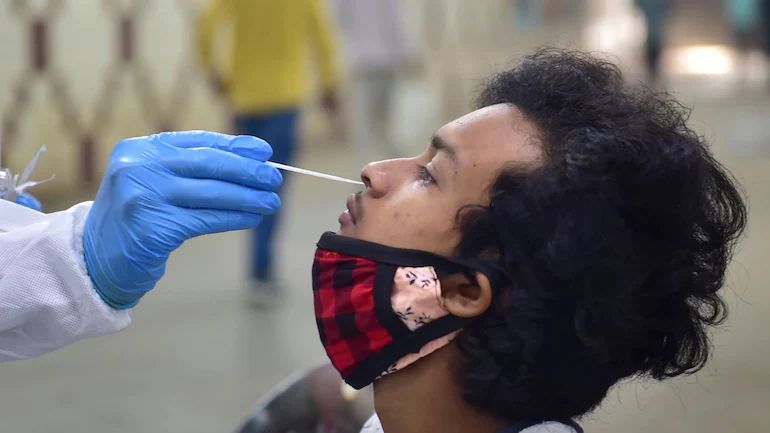
New Delhi: After reviewing all applications and data on antiviral pills, the Subject expert committee (SEC) a body under India’s drug regulator will be meeting today for antiviral pill Molnupiravir recommendations, sources told to ANI. According to the sources, almost 10 pharma companies have completed clinical trials of antiviral pills. The antiviral pill has shown a 30 percent reduction in hospitalisation and will be useful in the treatment of Covid-19. The SEC will review the Serum Institute of India’s Covovax, the WHO has recently granted emergency use listening (EUL) to Covovax. The US-based Novovax and Serum Institute of India has already received EUA for these vaccines in the Philippines. The SEC will also review data of the Corbevax vaccine which is produced by Hyderabad based pharmaceutical firm Biological E. The company has submitted phase-3 trials data of Corbevax and seeking EUA for same. The centre has already made an advance payment of 1500 crore to reserve 30 crores of Corbevax in August. Read all the Latest News , Trending News , Cricket News , Bollywood News , India News and Entertainment News here. Follow us on Facebook, Twitter and Instagram.
The SEC will also review data of the Corbevax vaccine, which is produced by Hyderabad-based pharmaceutical firm Biological E. The company has submitted phase-3 trials data of Corbevax and seeking EUA for same
Advertisement
End of Article


)

)
)
)
)
)
)
)
)



