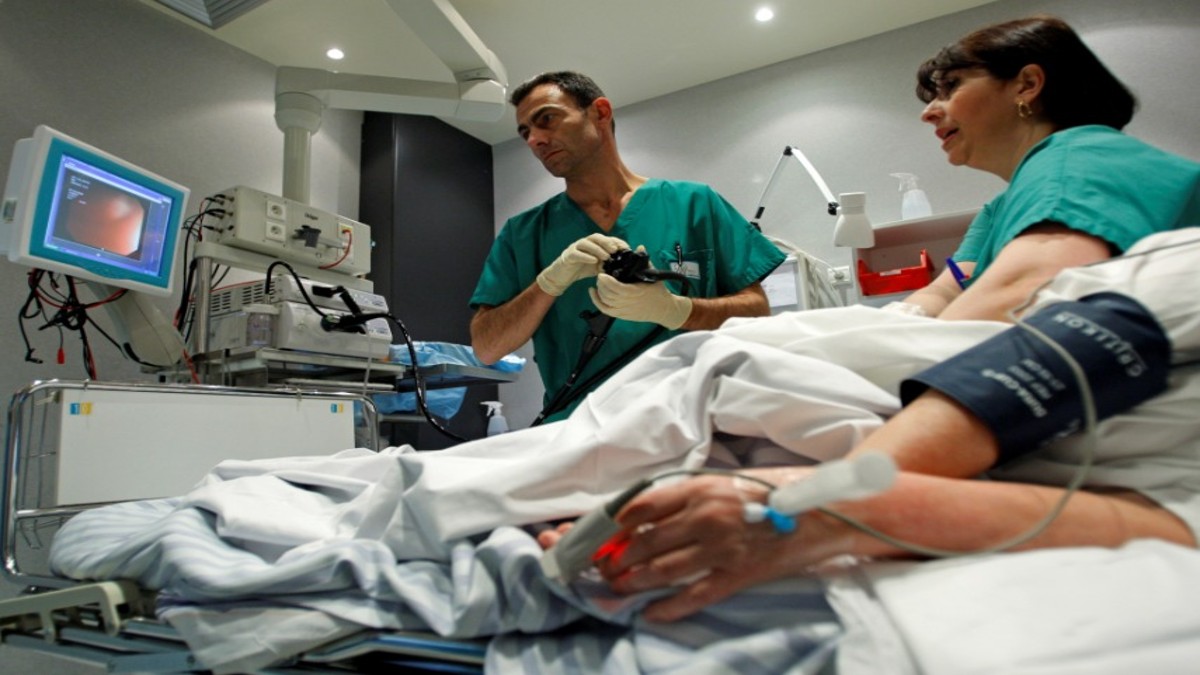
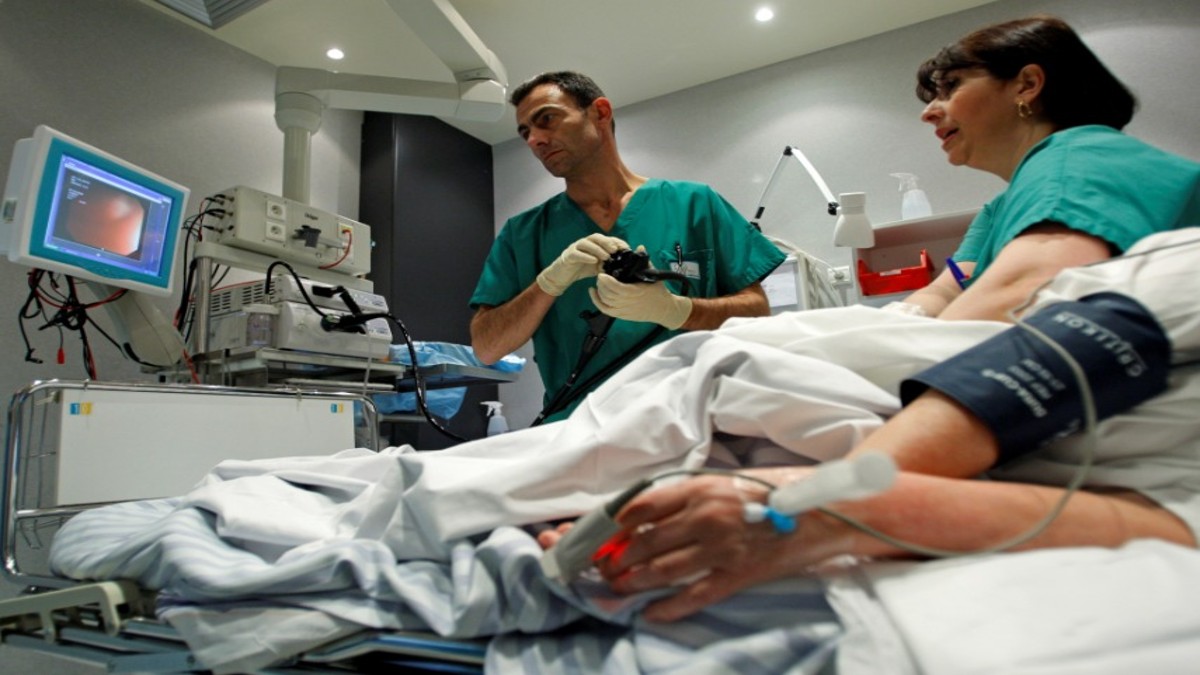
Ongoing research at IIT Bombay may pave the way for CAR T-cell therapy to become a possible treatment for cancer patients in India. Professor Rahul Purwar of the Department of Biosciences and Bioengineering launched the startup ImmunoACT four years ago to address the unavailability of the therapy in India. CAR T-cell therapy is available in the US from pharmacological giants Novartis and Gilead, but the costs involved are massive at Rs 3-4 crores per treatment. The researchers at IIT-B, in collaboration with Tata Memorial Hospital, have developed a therapy that will be much cheaper. According to estimates, treatment costs would be comparatively low at Rs 15 lakhs.
CAR T-cell therapy and how it works
Chimeric Antigen Receptor T-cell therapy (CAR T) is a new form of cancer immunotherapy wherein the body’s T cells (which are white blood cells involved in the body’s immune response) are modified by gene insertion to recognize and attack cancer cells. Here is a simplified version of the process:
- Extracting the T-cells: the patient is hooked to an apheresis machine which filters the blood and extracts T-cells from it. The blood is then returned to the body.
- T-cell modification: the antibody CAR is inserted into the DNA of the T-cell. This antibody is responsible for the immune response that targets specific types of cancer.
- Cell multiplication: the CAR T-cells are allowed to multiply in a lab until they are in the millions and then frozen. The patient may be given chemotherapy while this part of the process takes place.
- Modified cell re-introduction: the modified cells are then pumped back into the bloodstream and act as ‘attacker’ cells by targeting the cancer cells in the body.
Suitable candidates for CAR T-cell therapy
Called a ‘living drug’, the cells remain in the body and replicate, and if successful, kill off malignant cancer cells. The therapy has thus far been used for various types of leukaemia (blood cancer) and certain types of non-Hodgkin’s lymphoma. Research is ongoing on cancers of the cervix, mouth, throat and lung, among others.
CAR T-cell therapy is recommended for those cases where the response to other treatments has been limited - it is not yet a preferred treatment. Based on extrapolations from various cancer hospitals, the treatment will affect roughly 20,000 to 40,000 people suffering from leukaemia and lymphoma per year.
Prognosis and side effects
Initial results, particularly with leukaemia, have been encouraging. About 80% of those who received treatment went into remission. While this is an exciting development, research on the therapy is still limited and further work needs to be done to understand its potential and limitations.
Approximately 40-60% of those who undergo treatment have side effects. They are not usually serious, but can on occasion turn life-threatening especially within the first week of treatment. They include flu-like symptoms such as headaches, nausea and joint pain. There can also be neurological symptoms such as seizures, short-term memory loss and cardiovascular issues such as elevated blood pressure.
Tackling the expensive cost of CAR T-cell therapy
“The molecular structure of our product is different,” Professor Purwar explained over the phone. ImmunoACT has two patents pending on their technology; they have developed a CAR T-cell platform that attacks CD19+ B-cell malignancies which are expressed in certain types of leukaemia.
“Manufacturing costs are extremely high with this form of cell therapy. More than half of the cost goes to skilled manpower. The processes involved are highly technical and complex, and investments are risky. High costs prevail,” Prof. Purwar said.
Manufacturing costs will be lower in the startup’s model as scientists will be trained on the platform. Additionally, the new patent-pending technology will also lower costs. Lab work conducted so far has suggested that a single dose may cause a sufficient immune response.
IIT-B hopes to start clinical trials next year. This first requires getting permission from the Drug Controller General of India (DCGI), however.
Another player in the field
Bangalore based Immuneel Therapeutics, owned by Kiran Mazumdar’s Biocon has also entered the race to harness CAR T-cell technology and make it readily available. Partnered with the Pulitzer Prize-winning author Siddhartha Mukherjee, the business model of the venture is different from ImmunoACT’s. Immuneel will in-license the technologies and cut costs by streamlining the engineering and delivery process. The company will provide training to hospitals and provide the therapy in a fee-for-service model. Mazumdar says that the company is not committed to any particular technology; it is more about delivering the service cost-effectively and efficiently. The company hopes to infuse its first patient by September next year.
There are many reasons to be excited about the emerging field of immunotherapy in the country. The involvement of startups and the push to make the technology native will hopefully make the therapy available to those who need it. Cancer burden in the country is increasing and these advances will play a key role in saving lives.
For more information, please read our article on Cancer: Types, Stages, Risk Factors and Treatment _._
Health articles in Firstpost are written by myUpchar.com, India’s first and biggest resource for verified medical information. At myUpchar, researchers and journalists work with doctors to bring you information on all things health.


)




)
)
)
)
)
)
)
)