India’s COVID-19 vaccination drive is about to witness a change after the drug regulator on Thursday granted a conditional market authorisation for Covishield and Covaxin , the two coronavirus vaccines widely used in the country. We take a look at what is conditional market authorisation and how this affects the COVID-19 vaccine drive. What does conditional market authorisation mean? To date, Serum Institute of India’s Covishield and Bharat Biotech’s Covaxin has been available under ‘Emergency Use Authorisation’. Through the EUA route, the vaccines are available only in emergency situations; for instance in a public health emergency like the pandemic. In this case, the drug regulator, based on initial data from phase 3 of clinical trials, permits the use of vaccines. According to officials, as the two vaccines now meet the high standards of safety, effectiveness, and manufacturing quality that the Drugs and Cosmetics Act, 1940 requires of a new vaccine, they have been upgraded to “conditional market authorisation”. One must note that it is ‘conditional’, which means there are still some stipulations that need to be met. [caption id=“attachment_10326961” align=“alignnone” width=“640”] 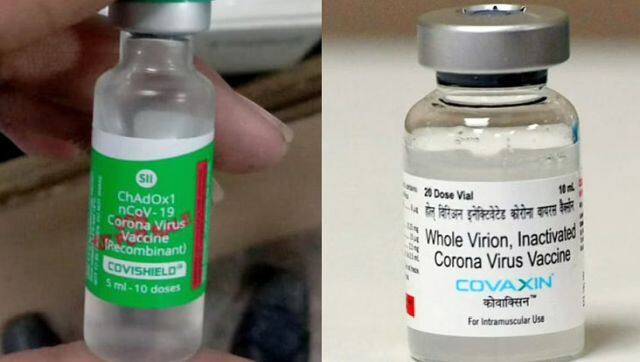 The Subject Expert Committee of the Central Drugs Standard Standard Control Oraganisation had on 19 January recommended upgrading the Covishield and Covaxin vaccines. ANI[/caption] What are these conditions? The approval is an upgrade from the emergency use authorisation and largely represents a regulatory endorsement of the safety doses. Soon after approval was granted, Health Minister Mansukh Mandaviya tweeted, “The @CDSCO_INDIA_INF has now upgraded the permission for Covaxin and Covishield from restricted use in emergency situations to normal new drug permission in the adult population with certain conditions.”
The @CDSCO_INDIA_INF has now upgraded the permission for COVAXIN and Covishield from restricted use in emergency situations to normal new drug permission in the adult population with certain conditions.
— Dr Mansukh Mandaviya (@mansukhmandviya) January 27, 2022
So, what are these conditions? According to officials, as per a PTI report, the firms shall submit data of ongoing clinical trials and the vaccines to be supplied for programmatic setting besides recording all vaccinations done within the country on the CoWIN platform. Adverse event following immunisation will continue to be monitored as part of the conditions. The firms will also have to submit data of overseas ongoing clinical trials of the product with due analysis. The conditional market authorisation came after the Subject Expert Committee of the Central Drugs Standard Standard Control Oraganisation had on 19 January recommended upgrading the Covishield and Covaxin vaccines. It advised grant of new drug status to the two vaccines from Emergency Use Authorisation permission granted earlier. So, what changes? As of now, nothing much changes. A person aware of the decision said the approval does not mean the vaccines can be sold over-the-counter by pharmacies, in the way that fully approved doses are. “Conditional access does not permit that; in fact, people wanting to take the shot will still have to approach a COVID vaccination centre. Pharmacies will not be able to stock it just yet,” a senior government official was quoted as saying to Hindustan Times. However, highly-placed sources said to NDTV that in the future to come, the two vaccines will be available in private clinics and hospitals at pre-decided MRP and people can buy those. Gradually, the vaccine will be available in the market at a price lower than at what it was being sold earlier at private hospitals, sources added. However, the sources said, as and when a private hospital or clinic administers a dose, it will still have to capture it on CoWin, the government’s digital vaccination platform. News agency PTI quoted a source as saying, “The idea is that those who are keen to get a booster dose can now avail it through private clinics as the vaccine available in private clinics can be used for any purpose, whether it’s first dose, second dose or a booster as one wishes.” Coronavirus in India On Friday, 2,51,209 people tested positive for coronavirus in a day, taking India’s total tally to over 4.06 crore, according to the Union Health Ministry data updated on Friday. In the same time period, the death toll has climbed to 4,92,327 with 627 fatalities reported. The daily positivity rate was recorded at 15.88 per cent, while the weekly positivity rate was 17.47 percent. Meanwhile, the cumulative number of anti-COVID vaccine doses administered in the country so far has crossed 164.44 crore. With inputs from agencies Read all the Latest News , Trending News , Cricket News , Bollywood News , India News and Entertainment News here. Follow us on Facebook, Twitter and Instagram.


)

)
)
)
)
)
)
)
)



