Scientists have long suspected that antibodies in the blood of COVID-19 patients that have successfully fought the infection and recovered could provide powerful protection against SARS-CoV-2 – the coronavirus that causes COVID-19. A recently published study has offered some evidence to back this theory with
successful tests in animals and human cell cultures. The study, published 16 June in
Science, shows a range of swift reactions to the deadly viral pandemic – setting the stage for upcoming clinical trials and additional tests of these antibodies. They are now being produced as a potential treatment and preventive course for the novel coronavirus infection. “The discovery of these very potent antibodies represents an extremely rapid response to a totally new pathogen,” co-senior author Dennis Burton, the James and Jessie Minor Chair in Immunology in the Department of Immunology & Microbiology at Scripps Research Institute,
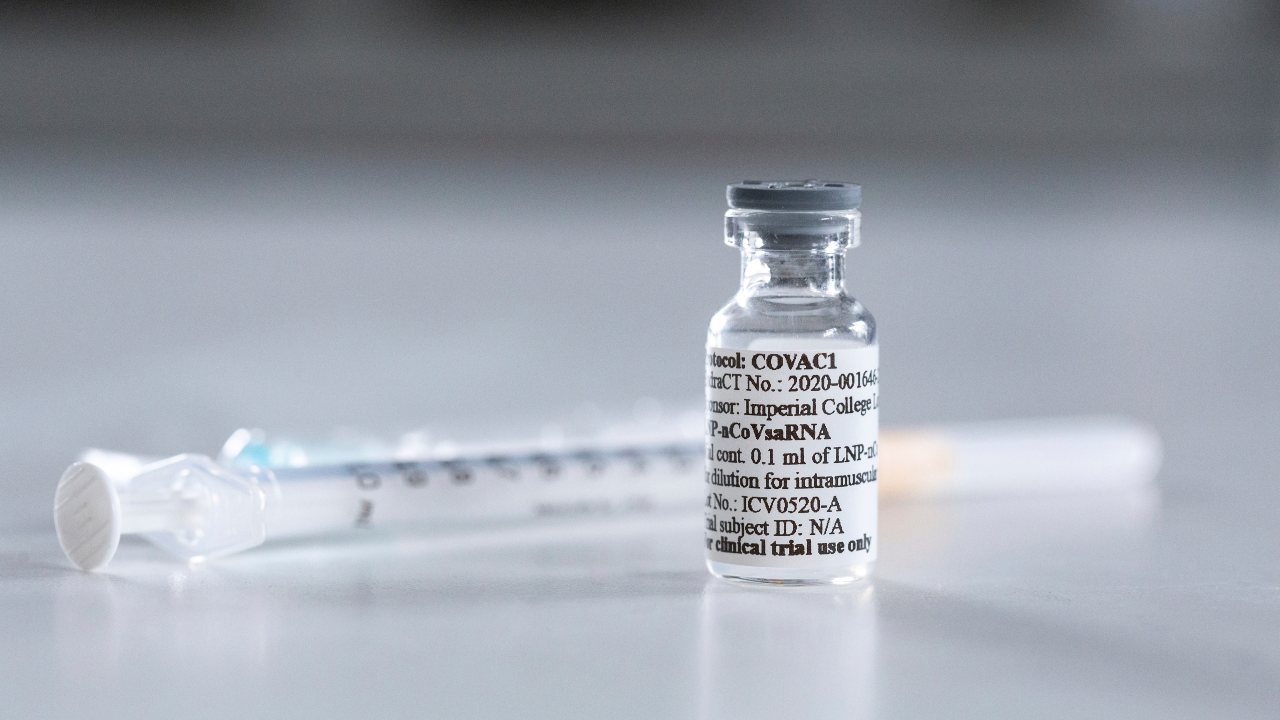
told SRI press. [caption id=“attachment_8488691” align=“alignnone” width=“1280”] A vaccine candidate against COVID-19 (the SARS-CoV-2 virus), provided by Imperial College London. About a dozen vaccine candidates are in early stages of testing in thousands of people. Image: Imperial College London via AP[/caption] In theory, if these antibodies were injected into patients in the early stages of COVID-19, it would reduce the level of virus and offer protection against the disease progressing in seriousness. The antibodies could potentially also offer vaccine-like protection for a limited period of time against SARS-CoV-2 infection for those most at-risk: healthcare workers, elderly people, others who haven’t responded well to traditional vaccines or those thought to have been recently exposed to the coronavirus. There are already many
**plasma therapy studies underway** in hospitals and clinics around the world. “It has been a tremendous collaborative effort, and we’re now focused on making large quantities of these promising antibodies for clinical trials,” co-lead author of the study Thomas Rogers, an adjunct assistant professor in the Department of Immunology & Microbiology at Scripps Research, and assistant professor of Medicine at UC San Diego, told SRI press. Also Read:
Can plasma therapy help people who have been exposed to COVID-19, stave off the infection? As of 16 June, over
8 million people have tested positive for SARS-CoV-2, resulting in the deaths of over
4,37,000 people worldwide. New cases continue to crop up every day – including in countries like New Zealand that were thought to be completely free of the coronavirus till the travel ban was lifted. Developing a treatment or vaccine for severe COVID-19 is currently the world’s top public health priority. One of the key approaches to new viral threats is identifying antibodies to the virus found in the blood of recovering patients, antibodies that neutralize the virus’s ability to infect cells. With modern biotechnology, these antibodies can then be mass-produced as a treatment that blocks severe disease and as a vaccine-like preventive that circulates in the blood for a few weeks offering protection against infection. [caption id=“attachment_8488131” align=“alignnone” width=“1280”]
An illustration of the 2019 Novel Coronavirus (2019-nCoV), which was identified as the cause of an outbreak of respiratory illness first detected in Wuhan, China and now a raging global pandemic. Image: CDC[/caption] This approach already has been demonstrated successfully in the past against deadly viruses like the Ebola virus and the pneumonia-causing respiratory syncytial virus (RSV). In the ongoing project, researchers at UC San Diego took blood samples from patients who had recovered from mild-to-severe cases of COVID-19. In tandem, researchers at Scripps Research and IAVI developed a series of test cells that express the human ACE2 receptor – which SARS-CoV-2 uses to get into human cells. The team tested whether antibody-containing blood from the patients could inactive the virus by binding to it and strongly block it from infecting these test cells. They managed to pick out over 1,000 unique antibody-producing immune cells (called B cells) – each of which produced a unique antibody against SARS-CoV-2. Using tools in genetics, the team recovered the gene sequences from these B cells so that they could produce the antibodies in a laboratory. As they screened this large set of potential antibodies for SARS-CoV-2, they identified many that could block the virus in test cells, even in tiny quantities. One of the antibodies could even protect hamsters against a heavy dose of exposure to COVID-19, as per
the report. If further safety testing in animals and clinical trials in humans go well, the antibodies could be used in clinical settings as early as next January, the researchers say. “We intend to make them available to those who need them most, including people in low- and middle-income countries,” Landais says. Over the course of their research, the team also isolated an antibody from COVID-19 patients that can neutralize SARS-CoV – the coronavirus relative responsible for a massive outbreak of severe acute respiratory syndrome (SARS) in Asia in 2002-2004. “That discovery gives us hope that we will eventually find broadly neutralizing antibodies that provide at least partial protection against all or most SARS coronaviruses, which should be useful if another one jumps to humans,” Burton said.
Developing a treatment or vaccine for severe COVID-19 is currently the world’s top public health priority.
Advertisement
End of Article


)

)
)
)
)
)
)
)
)



