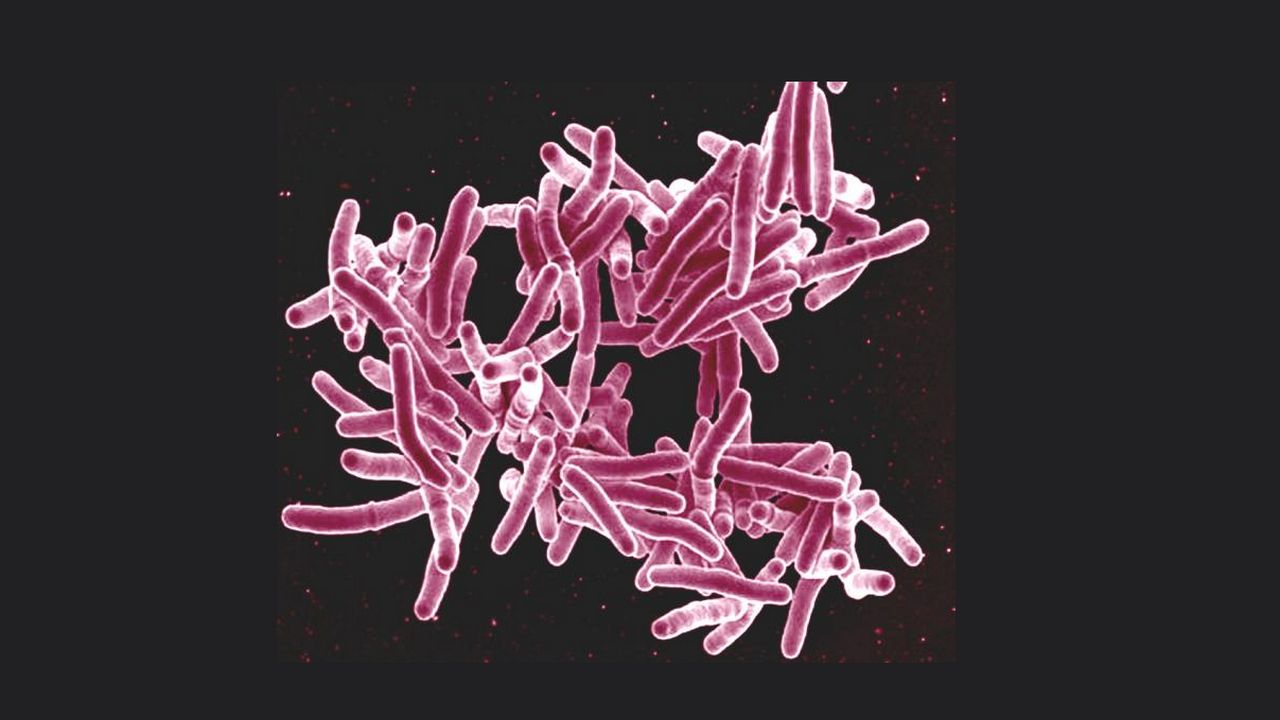
Tuberculosis infection results in approximately nine million new cases worldwide every year. The TB-causing bacterium can remain dormant in the human body for a long time, even for several decades before it becomes infectious. The bacterium remains ensconced within a type of white blood cells called macrophages when it is in its latent form. A team of researchers from Kolkata-based CSIR-Indian Institute of Chemical Biology, Bose Institute and Jadavpur University have figured out how tuberculosis bacterium is released from its reservoir inside the human body. A macrophage, in fact, is an important part of the immune system. The word ‘macrophage’ literally means a ‘big eater’. It is an amoeba-like organism and its job is to clean the body of microscopic debris and invaders. It has an innate ability to locate and consume invaders such as bacteria, viruses, fungi, and parasites. However, the story is different with TB bacterium. Instead of killing it, the macrophage creates a sac-like formation called granuloma around it. Granuloma keeps the bacillus contained and under control. The equilibrium can last for even several decades until it gets broken leading to the release of infectious bacteria into the human body. This can happen due to several reasons such as lowered immunity because of physical weakness or infections such as HIV. [caption id=“attachment_7003001” align=“alignnone” width=“1280”] Representational image. Image Credit: NIAID/Flickr[/caption] Scientists globally have been trying to figure out the molecular mechanism of the release of infectious TB bacteria from the granuloma. The new study by Kolkata researchers could fill this knowledge gap. The team has discovered that a protein molecule called MPT63 secreted by the bacteria may be playing a role in the release process. Synthetically produced MPT63 protein molecule was subjected to different levels of acidity and it was found that when the acidity value is high its structure changed dramatically. It started as what is called a beta-sheet and it turned into a helical form in the acidic condition, which prevails in matured granuloma. The protein, which was found to have no apparent function in the folded beta-sheet, suddenly became toxic on assuming the helical form, and got into the cell membrane, leading to the formation of pores and consequently the death of the host cells and release of the bacteria. “Our team would now try to validate these findings in field strains of TB bacillus and see whether they can be used to develop new therapeutic interventions,” said Dr Krishnananda Chattopadhyay, Head of Structural Biology and Bioinformatics Division at IICB and team leader, while speaking to India Science Wire. Besides him, the team consisted of Achinta Sannigrahi, Indrani Nandi, Sayantani Chall, Junaid Jibran Jawed, and Animesh. Halder, Subrata Majumdar, Sanat Karmakar. The study results will soon be published in journal ACS Chemical Biology.
A protein molecule called MPT63 secreted by the bacteria maybe playing a role in the release process.
Advertisement
End of Article


)
)
)
)
)
)
)
)
)



