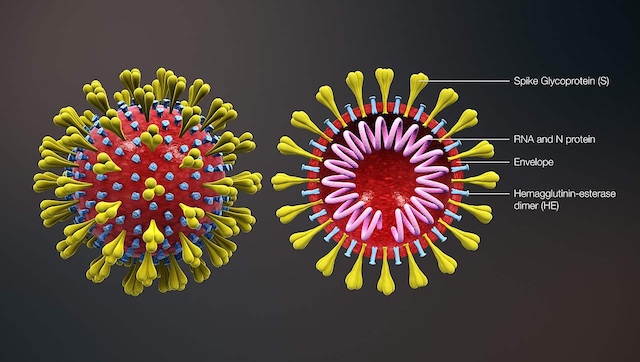
Patients who recover from the COVID-19 infection have antibodies against the virus in their body. These antibodies can help in faster recovery if given to an active patient of COVID-19 infection. The diagnostic tests that look for antibodies in recovered COVID-19 patients only detect the presence of antibodies and do not check for their ability to block the virus from binding to human cells. Though there are some tests that can check the effectiveness of the antibodies against the virus, they are quite expensive and require longer to process and give results. Given the current situation, it has become vital to find a technique that could help provide this testing facility in a community setting while overcoming the barriers. As per a recent study published in The Journal of Infectious Diseases on 23 October, 2020, it was found that with the help of microsphere-based flow cytometry technique, scientists would not only determine the presence of antibodies but would also the effectiveness of those antibodies. The new antibody detection technique It is a known fact that the SARS-CoV-2 enters the human body with the help of spike proteins present on its surface that bind to the ACE2 protein present on the surface of human cells. To fight the virus, the human immune system forms different types of antibodies against it. Neutralising antibodies block the virus from binding to human cells, thus restricting the spread of the infection. Currently, the diagnostic tests determine the performance of immunity by collecting antibody-rich blood from recovered patients, mixing it with the virus and then exposing it to live cells. It takes three days for the test to give results. However, a study conducted by researchers from Seattle Children’s Research Institute, USA, found that with the help of a test called immunoprecipitation detected by flow cytometry (IP-FCM), the strength of the immune response can be determined overnight. For this study, the scientists collected blood samples of 24 patients who tested positive for COVID-19 infection, presented with mild to moderate disease and were not hospitalised. Of these 24, 12 patients experienced fever while 16 experienced cough. These samples were compared with those of 30 healthy people who were kept in the control group. The samples were tested using IP-FCM. Both COVID-19 positive and negative blood samples were initially heated at 56°C for 1 hour and then spun at 13,000g for 10 minutes at 4°C. These samples then went under the process of dilution and buffer. Results of the study The results of the study showed that 94 percent of the 24 COVID-19 positive patients had antibodies against the novel coronavirus by just over a month (on average) post-infection. It was also found that their antibodies were quite effective at neutralising the binding between the spike protein and the ACE2 receptors present on cells. Scientists further stated that participants who presented with fever had high levels of antibodies in their body as compared to those who got a cough. Conclusion The study concluded that immunoprecipitation detected by flow cytometry (IP-FCM) test can be used as an effective tool to determine the ability of antibodies to inhibit and block the virus from binding to cells. The scientists added that with this newly developed diagnostic test, they would be able to test the presence of convalescent plasma in those who have recovered from COVID-19 on a larger scale. This test would also allow them to be able to assess the response of vaccine (once developed) in their body. The scientists are also using this technique to determine the effectiveness of various drugs that may restrict the binding between ACE2 receptors and the spike protein of the virus. For more information, read our article on Convalescent plasma therapy. Health articles in Firstpost are written by myUpchar.com, India’s first and biggest resource for verified medical information. At myUpchar, researchers and journalists work with doctors to bring you information on all things health.
For this study, the scientists collected blood samples of 24 patients who tested positive for COVID-19 infection, presented with mild to moderate disease and were not hospitalised. Out of these 24, 12 patients experienced fever while 16 experienced cough
Advertisement
End of Article


)

)
)
)
)
)
)
)
)



