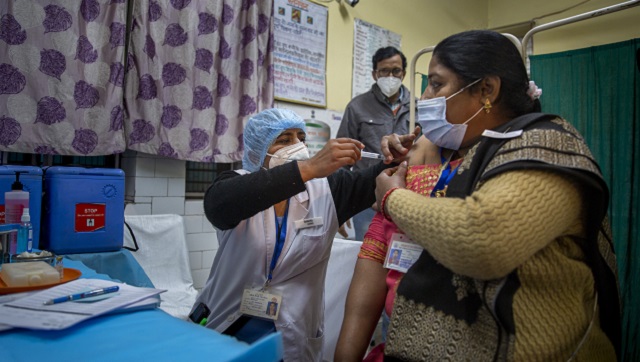
A COVID-19 expert panel of the CDSCO on Saturday recommended a second COVID-19 vaccine COVAXIN, indigenously developed by Bharat Biotech and the ICMR, for emergency use authorisation, especially against the mutant strains. The SEC’s recommendations will now be considered by the Drugs Controller General of India (DCGI), who will grant the final approval, reports said. This came on a day India tested its COVID-19 vaccine delivery system with a nationwide trail, as it prepares to roll out an inoculation programme to stem the coronavirus pandemic. Meanwhile, the number of new COVID-19 cases in the country fell below 20,000 while the national recovery rate rose to 96.12 percent, according to data from the Union Ministry of Health and Family Welfare. Additionally, Union health minister Harsh Vardhan appealed to people not to be “misguided” by rumours and disinformation regarding the safety and efficacy of the vaccine, while announcing that the govt would bear the cost towards inoculating one crore healthcare workers and two crore frontline workers. Also, the ICMR said that India has successfully cultured the new coronavirus strain, which originated in the UK. In a tweet, the ICMR claimed that no country has yet reported successful isolation and culture of the UK-variant of SARS-CoV-2. Culture is the process by which cells are grown under controlled conditions, generally outside their natural environment. COVAXIN recommended for emergency use approval Day after clearing the Serum Institute of India’s emergency use authorisation application for the Oxford-AstraZeneca vaccine ‘Covishield’, the Subject Expert Committee on COVID-19 of the Central Drugs Standard Control Organisation (CDSCO) Saturday issued a similar direction for Bharat Biotech’s COVAXIN, paving the way for the roll-out of COVID-19 shots in the country in the next few days. Both the vaccines now await the final approval of the Drugs Controller General of India (DCGI), the Union health ministry said on Saturday. COVAXIN has been indigenously developed by Bharat Biotech in collaboration with the Indian Council of Medical Research (ICMR). The SEC has recommended “grant of permission for restricted use in emergency situation in public interest as an abundant precaution, in clinical trial mode, especially in the context of infection by mutant strains, to Bharat Biotech,” the health ministry stated. However, PTI quoted sources as saying that the committee also stated that Bharat Biotech shall continue the ongoing phase 3 clinical trial and submit data emerging from the trial as and when available. The committee noted that the vaccine is an inactivated whole virion, coronavirus vaccine, having the potential to target mutated coronavirus strains, the PTI report added, citing sources. The data generated so far demonstrates a strong immune response (both antibody as well as T cell) and in-vitro viral neutralisation. After detailed deliberations, the SEC has also recommended for grant of permission to Cadila Healthcare Ltd in Ahmedabad for the conduct of phase-3 clinical trial protocol for its vaccine candidate. Vaccine dry run held across India As India came closer to the roll-out of COVID-19 vaccine, dry runs were held across the country on Saturday. The dry-run was being conducted in state capitals in at least three session sites. Some states also included districts that are situated in difficult terrains or have poor logistical support, PTI reported. Maharashtra, Kerala and Gujarat conducted the mock drill in four districts each, Karnataka and Tamil Nadu in five districts each, while Rajasthan carried out the dry-run in seven districts. The first phase of the dry run was conducted in Andhra Pradesh, Assam, Gujarat, Punjab on 28 and 29 December, with at least 125 intended beneficiaries at five sites in a maximum of two districts each. The Union health ministry had said that no major issues were observed during the pilot dry run and all states had expressed confidence in the operational guidelines and IT platform for large-scale programme implementation. Specific teams were formed for various tasks conducted on Saturday’s rehearsal by the district administrations. Activities like uploading of dummy beneficiary data, session site creation, vaccine allocation, communicating vaccination details to beneficiaries and vaccinators, and beneficiary mobilisation, etc, were carried out. In Delhi, where the dry run was held at three sites, state health minister Satyendar Jain said that free coronavirus vaccine will be provided to the people in the city. Interacting with reporters during a visit to a facility in Daryaganj, he said the system “seems flawless” as of now. On Saturday, the dry run was conducted at identified health centres in Nagpur, Jalna, Pune and Nandurbar districts of Maharashtra. In Kerala, at least 25 health workers each in four districts — Thiruvananthapuram, Idukki, Wayanad and Palakkad — took part in the drill. In West Bengal, the dry run was held at Urban Primary Health Centres in Duttabad and Madhyagram and Amdanga Rural Hospital in North 24 Parganas district A senior official in Assam said the dry run will continue regularly in the state till actual vaccination takes place. In Karnataka, the exercise was held at Kalaburagi, Shivamogga, Mysuru, Belagavi, and Bengaluru districts, and in Tamil Nadu, the dry run was held at three centres each in the districts of Chennai, Tiruvallur, Nilgiris and Tirunelveli, and five in Coimbatore. An important focus of the dry run was on the management of any possible adverse events following immunisation (AEFI), and adherence and management of infection control practices at the session site to prevent disease transmission, the union health ministry had said. The states and UTs were asked to ensure that the data of beneficiaries is uploaded in the Co-WIN app, ensure physical verification of all proposed sites for the adequacy of space, logistical arrangements, internet connectivity, electricity, safety, etc. and prepare at least three model session sites in each state (at state capital) for demonstration. They were asked to ensure that the model sites have separate entry and exit in a ’three-room set-up’ with adequate space outside for awareness generation activities. The dry run will equip the state and UT administration in the management of vaccine supply, storage and logistics including cold chain management, the ministry had stated. Won’t compromise any protocol in vaccine approval: Harsh Vardha Union Health Minister Vardhan said on Saturday free vaccine will be provided in the first phase to the most prioritised beneficiaries, including one crore healthcare and two crore frontline workers. He also said the details of how 27 crore priority beneficiaries — those above 50 years of age and those below it having comorbidities — will be vaccinated until July were being finalised. Vardhan stressed that there would be no compromise on any protocol while approving the vaccine. The minister dispelled doubts regarding the country’s capability to undertake such a massive vaccination drive, describing how India had unparalleled experience in dealing with immunisation and runs one of the largest such programmes in the world. “I appeal to the people not to be misguided by rumours regarding the safety and efficacy of the COVID-19 vaccine. We will not compromise on any protocol before approving a vaccine,” Vardhan said underlining that vaccine hesitancy was an issue even when the country started polio immunisation drive, but “we must remember its success”. The health minister has been credited with doing pioneering work in the successful mass pulse polio immunisation programme. “It is as a result of our steadfastness and dedication that India was declared polio-free in 2014. Our rich learning from the earlier immunisation drives including the Polio vaccination campaign is being used to guide our present countrywide COVID-19 vaccination campaign,” he was quoted as saying in a statement. Guidelines for the nationwide dry run were updated based on the feedback from the insightful pilot dry run held earlier in four states, Vardhan said. One primary contact tests positive for UK strain in Karnataka; 37 UK-returnees report infection in Kerala Amidst the nationwide trial of the COVID-19 vaccination system, the Central Government and various state governments continued to trace returnees from the UK who have tested positive, in a bid to curb the spread of the new ‘infectious’ virus strain detected in Britain. One primary contact of a UK returnee was among 10 people who tested positive for the new strain of the 2019 novel coronavirus and all of them are undergoing treatment, Karnataka health minister K Sudhakar said on Saturday. “Ten people have so far been confirmed with the new variant of coronavirus infection… nine of them are returnees from the UK, while one is a contact, the mother of one them. All of them are undergoing treatment and are without any serious health condition. They will recover soon,” he said. The minister told reporters here that so far 32 people who returned from the UK and 10 of their contacts — a total of 42 — have been confirmed with COVID-19 infection during the RT-PCR test. Their samples were sent for genomic sequencing and 10 among them were confirmed to be infected by the new variant of the virus. In Kerala, 37 people who returned from the UK to Kerala recently tested positive for COVID-19, state health minister KK Shailaja said. She added that the samples of the UK returnees have been sent to the National Institute of Virology (NIV) for further testing for the mutant variant. She said that of the 11 results received from NIV, none had tested positive for the new variant. However, in Gujarat, four COVID-19 positive people were confirmed to have been infected by the new strain, PTI reported. Results of 15 samples that had tested positive for coronavirus on arrival in Ahmedabad are currently pending with the NIV. “All the passengers who had reached Ahmedabad from the UK were screened, and the samples of those who had tested positive for the novel coronavirus were sent to the NIV. We have received intimation that the UK strain of coronavirus has been detected in four such cases,” Principal Secretary (Health), Jayanti Ravi told reporters. All UK passengers to be tested on arrival between 8-30 Jan; India-UK flights to begin from 6 Jan All passengers coming from the UK between 8 and 30 January would be subjected to self-paid COVID-19 tests on arrival, the health ministry said in a standard operating procedure (SOP) issued on Saturday while the civil aviation ministry announced the decision to resume flights between the two countries. Moreover, each passenger arriving from the UK would have to bring his or her COVID-19 negative report from a test done 72 hours prior to the journey, the SOP stated. “Airlines to ensure the availability of negative test report before allowing the passenger to board the flight,” the health ministry’s SOP stated. It said adequate arrangements should be made for the passengers who will be waiting for their RT-PCR test or its results at the airport. “Passengers testing positive shall be isolated in an institutional isolation facility in a separate (isolation) unit coordinated by the respective state health authorities,” the SOP said. If the genomic sequencing indicates the presence of the new variant of SARS-CoV-2, the patient will continue to remain in a separate isolation unit, it said. The patient will be tested on the 14th day after having tested positive and he or she will be kept in the isolation facility till his or her sample has tested negative, it added. Additionally, Union aviation minister Hardeep Singh Puri said that flights from India to the UK will resume from 6 January, while services from the UK to India will resume from 8 January onwards. “Resumption of flights between India & UK: India to UK from 6 Jan 2021. UK to India from 8 Jan 2021. 30 flights will operate every week. 15 each by Indian & UK carriers (sic),” Puri said on Twitter. “This schedule is valid till 23 Jan 2021. Further frequency will be determined after review (sic),” he added. Earlier, Puri had announced on Friday that only 30 flights per week will operate between India and the UK when services resume from 8 January and that this arrangement will continue till 23 January. COVID-19 caseload details India’s caseload rose to 1,03,05,788 on Saturday, while the number of people who have recuperated from the disease crossed 99 lakh, according to the health ministry. A total of 19,079 people tested positive for the coronavirus infection on Saturday, while the toll due to the disease climbed to 1,49,218 in the country with 224 new fatalities, the ministry’s data updated at 8 am showed. The number of active COVID-19 cases remained below three lakh for the 12th consecutive day. There are 2,50,183 active coronavirus cases in the country, which accounts for 2.43 percent of the total caseload, the data stated. According to the ICMR, 17,39,41,658 samples were tested for COVID-19 in the country till 1 January, including 8,29,964 on Friday. The 224 new fatalities include 59 from Maharashtra, 26 from West Bengal, 23 from Kerala and 21 from Delhi. Of the 1,49,218 COVID-19 deaths reported in the country so far, Maharashtra has accounted for the highest of 49,580, followed by Tamil Nadu (12,135), Karnataka (12,096), Delhi (10,557), West Bengal (9,738), Uttar Pradesh (8,379), Andhra Pradesh (7,108) and Punjab (5,349). With inputs from PTI
The recommendation for Bharat Biotech’s vaccine came a day after the COVID-19 expert panel cleared the Serum Institute of India’s emergency use authorisation application for the Oxford-AstraZeneca vaccine ‘Covishield’
Advertisement
End of Article


)

)
)
)
)
)
)
)
)



