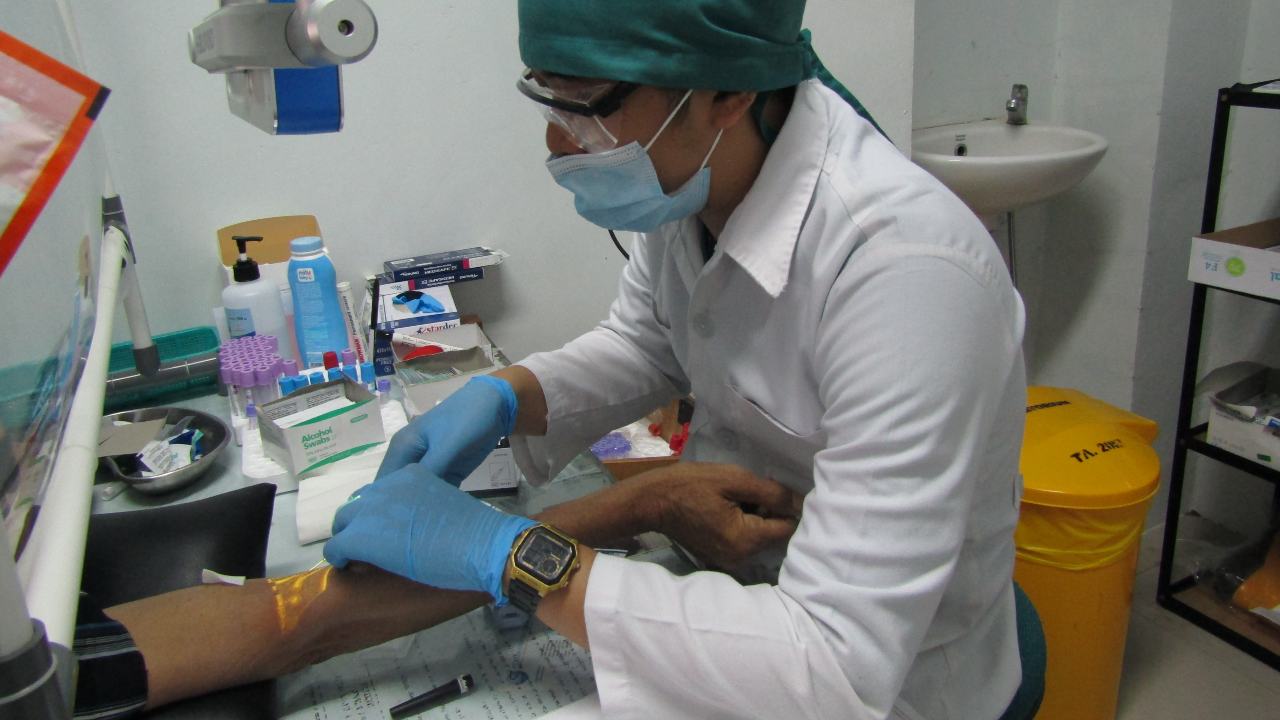
New data from Oxford University shows that its vaccine ChAdOx1 nCov-2019 produces a strong immune response in older adults, aged 56-69 and those over 70 years of age. This is an important milestone as this group is considered ‘high risk’ as they are the most vulnerable to the virus which is oftentimes fatal. With the vaccine showing the promise in producing immunity to people of this age group, it could be a potentially lifesaving tool in the pandemic. Dr Maheshi Ramasamy, Investigator at the Oxford Vaccine Group and Consultant Physician said in a statement, “Older adults are a priority group for COVID-19 vaccination because they are at increased risk of severe disease, but we know that they tend to have poorer vaccine responses.” According to researchers, during the Phase 2 trials, the vaccine showed similar immune responses across all the three age groups – 18-55, 56-69, and 70 and over. These results are also consistent with the Phase I data that showed immunity in the adults from ages 18 to 55. Ramasamy told The Guardian that the “robust antibody and T-cell responses” they saw in the group of older people were encouraging. “We were pleased to see that our vaccine was not only well-tolerated in older adults; it also stimulated similar immune responses to those seen in younger volunteers. The next step will be to see if this translates into protection from the disease itself,” he added. As per a press release on the early results, the vaccine was tested in 560 adults who received two doses of the vaccine or a placebo vaccine. They also did not have any serious adverse health events related to the vaccine in the volunteers. From the 560 volunteers, 240 of them were over the age of 70 years. According to a report by the BBC, two weeks after the volunteers were administered a second dose, more than 99 percent of them had neutralising antibody responses regardless of age. The study’s lead author, Andrew Pollard from the University of Oxford also told The Guardian, “Immune responses from vaccines are often lessened in older adults because the immune system gradually deteriorates with age, which also leaves older adults more susceptible to infections. As a result, it is crucial that Covid-19 vaccines are tested in this group who are also a priority group for immunisation.” The study also found that the vaccine was less likely to cause local reactions at the site where the injection was administered and symptoms on the day of vaccination in older adults than in the younger group. This new data comes from Phase 2 of the study and has been published in The Lancet journal. The Phase 3 trials of the vaccine are underway and early efficacy readings could come out in the upcoming weeks.
Two weeks after volunteers were administered a second dose, over 99 percent of them had neutralising antibody responses regardless of age.
Advertisement
End of Article


)
)
)
)
)
)
)
)
)



