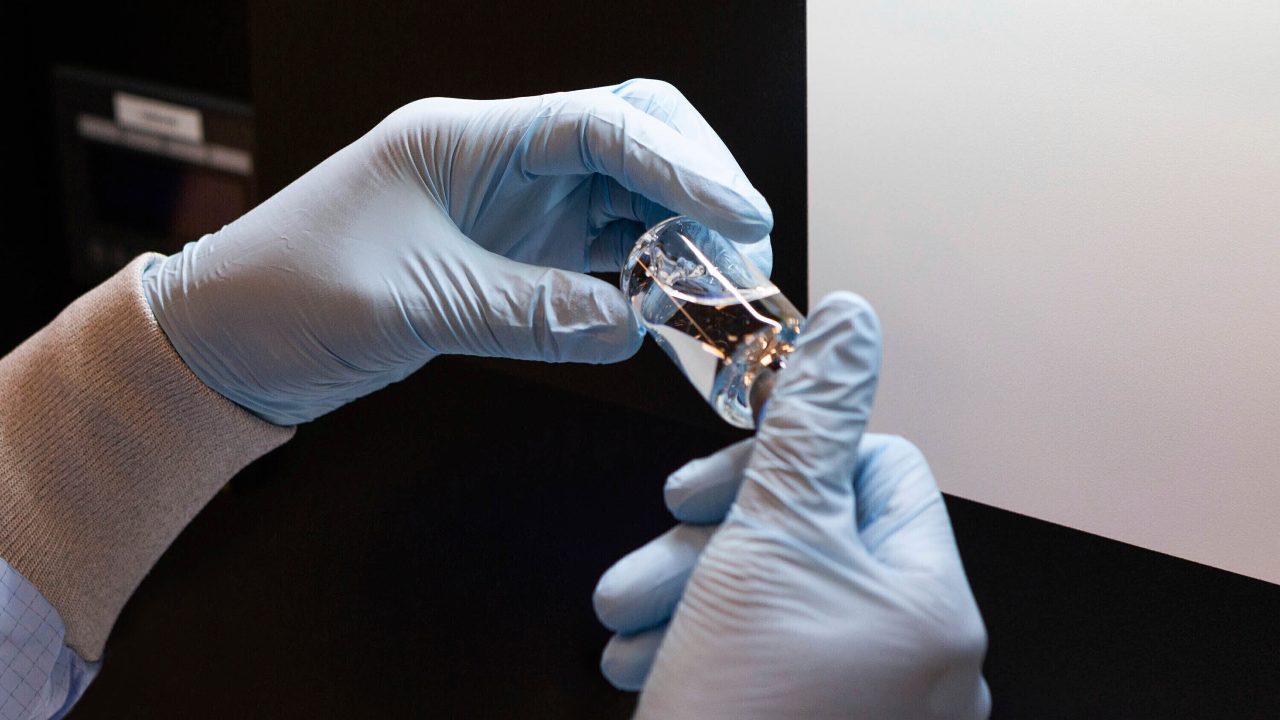
India’s drug regulator has granted US pharma giant Gilead Sciences marketing authorisation for its anti-viral drug remdesivir for “restricted emergency use” on hospitalised Covid-19 patients in view of the crisis posed by the pandemic. The approval process for remdesivir was accelerated in view of the emergency situation and the unmet need for medicines in light of the coronavirus outbreak, a source in the know of the developments told PTI. The drug has been allowed for restricted emergency use for the treatment of suspected or laboratory-confirmed cases of Covid-19 in adults and children hospitalised with severe symptoms, subject to several safeguards, the source said. [caption id=“attachment_8294201” align=“alignnone” width=“1280”] A vial of the investigational drug remdesivir is visually inspected at a Gilead manufacturing site in the United States. Given through an IV, the medication is designed to interfere with an enzyme that reproduces viral genetic material. Image credit: Gilead Sciences via AP[/caption] “The drug, which is administered in the form of an injection, has been approved to be sold by retail on the prescription of specialists for use in hospital or institutional setup only,” the source told PTI. “The approval process for remdesivir was accelerated by invoking special provisions under the New Drug and Clinical Trial Rules, 2019, which provides for waiver of clinical trials in special circumstances,” the source said. Gilead Sciences had on 29 May applied for
**marketing authorisation for remdesivir** in India. The drug is being touted as a potential treatment for Covid-19. The approval was given following consultation with the subject expert committee of the Central Drugs Standard Control Organisation (CDSCO). “The drug is given in the form of an injection and that is why it has to be administered under the supervision of a doctor in a hospital setting,” the source said. Gilead Sciences, the patent holder of the drug, has the complete data about the pre-clinical and clinical studies for remdesivir, the source added. The medicine has been issued an Emergency Use Authorization (EUA) by the United States Food and Drug Administration (FDA) to treat hospitalised coronavirus-infected patients. Meanwhile, the applications of two Indian pharmaceutical companies — Cipla and Hetero Labs — seeking permission to manufacture and sell remdesivir in India is still under consideration. Gilead Sciences Inc has entered into non-exclusive licensing agreements with pharma firms, including three domestic majors Cipla, Jubilant Life Sciences and Hetero, for manufacture and distribution of remdesivir. The Indian Council of Medical Research had earlier said anti-viral medication remdesivir, which was used during the Ebola outbreak, may inhibit the SARS-CoV-2 replication and research on its efficacy in the treatment of Covid-19 is a part of WHO’s ‘Solidarity Trial’. According to a recently published study in the New England Journal of Medicine, two out of three critically ill coronavirus patients who were on oxygen support showed signs of improvement when they were administered remdesivir. All new drugs have to undergo trials before getting approval for marketing them in India. But the New Drug and Clinical Trial Rules, 2019, provide for certain clauses, according to which the provision of waiver of local phase-III clinical trials of the drug is approved and marketed in certain countries (as notified from time to time) subject to certain conditions like national emergency or epidemics in the public interest.
India has allowed adults and children who have been hospitalised with severe COVID-19 symptoms to be treated with Remdesivir
Advertisement
End of Article


)

)
)
)
)
)
)
)
)



