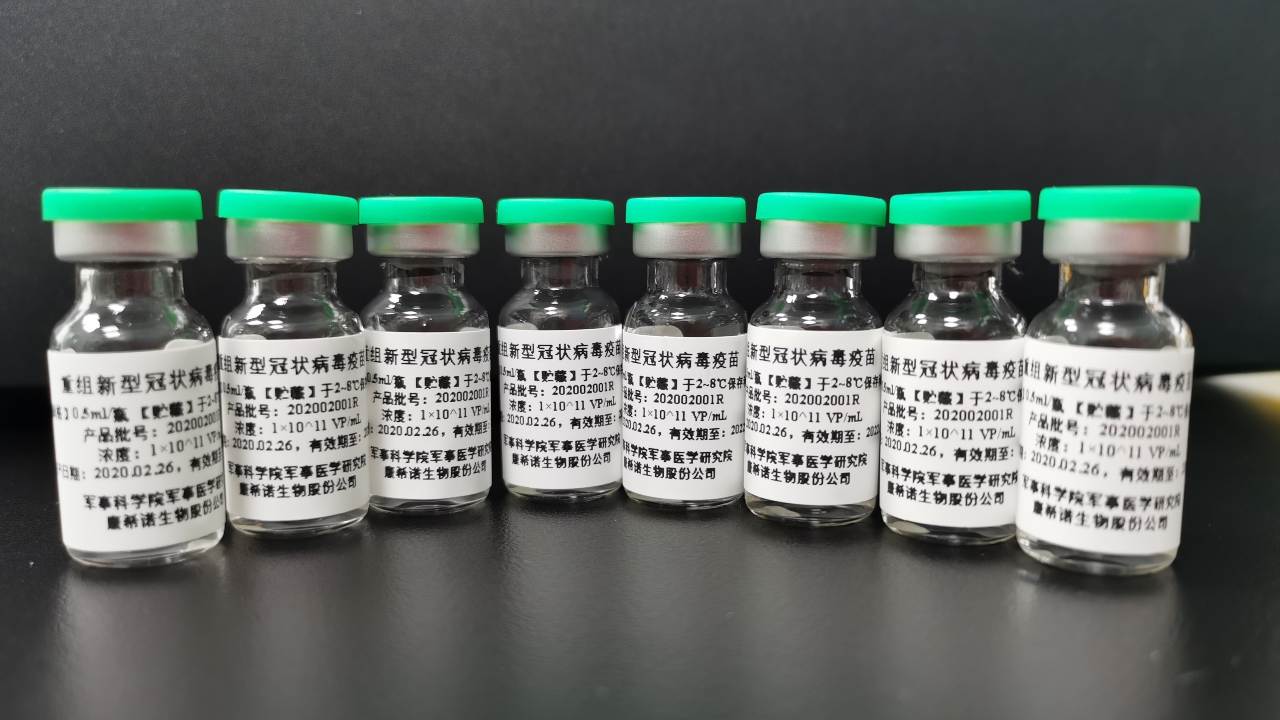
China’s military has received the green light to use a COVID-19 vaccine candidate developed by its research unit and CanSino Biologics after clinical trials proved it was safe and showed some efficacy, the company said on Monday. The Ad5-nCoV is one of China’s eight vaccine candidates approved for human trials at home and abroad for the respiratory disease caused by the new coronavirus. The shot also won approval for human testing in Canada. [caption id=“attachment_8544611” align=“alignnone” width=“1280”] he Ad5-nCoV is one of China’s eight vaccine candidates approved for human trials at home and abroad for the respiratory disease caused by the new coronavirus. Image credit: CanSino Biologicals[/caption] China’s Central Military Commission approved the use of the vaccine by the military on 25 June for a period of one year, CanSino said in a filing. The vaccine candidate was developed jointly by CanSino and a research institute at the Academy of Military Science (AMS). “The Ad5-nCoV is currently limited to military use only and its use cannot be expanded to a broader vaccination range without the approval of the Logistics Support Department,” CanSino said, referring to the Central Military Commission department which approved the military use of the vaccine. CanSino declined to disclose whether the inoculation of the vaccine candidate is mandatory or optional, citing commercial secrets, in an email to Reuters. The military approval follows China’s decision earlier this month to offer two other vaccine candidates to employees at state-owned firms travelling overseas. The Phase 1 and 2 clinical trials of the CanSino’s vaccine candidate showed it has the potential to prevent diseases caused by the coronavirus, which has killed half a million people globally, but its commercial success cannot be guaranteed, the company said. Separately, AMS received an approval earlier this month to test its second experimental coronavirus vaccine in humans. No vaccine has yet been approved for commercial use against the illness caused by the new coronavirus, but over a dozen vaccines from more than 100 candidates globally are being tested in humans.
The vaccine has been approved for a period of one year after clinical trials proved it was safe and showed some efficacy.
Advertisement
End of Article


)

)
)
)
)
)
)
)
)



